Ahh!!! The Devil's Breath Scopolamine Angel's Trumpet
They use ordinary people to do there crimes. That's what I think is really going on out here.
ate 25 years of humanity at its best.
Contents
hide
Tropane alkaloid
Tools
Appearance
hide
Text
Small
Standard
Large
Width
Standard
Wide
Color (beta)
Automatic
Light
Dark
From Wikipedia, the free encyclopedia
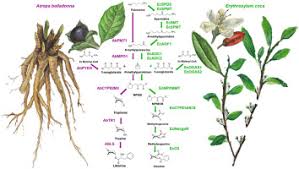
Chemical structure of tropane which forms the core of tropane alkaloidsChemical structure and phylogeny of tropane alkaloids. Displayed are 3 chemical compounds that occur as natural products in 5 plant species
Tropane alkaloids are a class of bicyclic [3.2.1] alkaloids and secondary metabolites that contain a tropane ring in their chemical structure.[1] Tropane alkaloids occur naturally in many members of the plant family Solanaceae. Certain tropane alkaloids such as cocaine and scopolamine are notorious for their psychoactive effects, related usage and cultural associations.[2][3][4][5][6][7] Particular tropane alkaloids such as these have pharmacological properties and can act as anticholinergics or stimulants.
Classification
Anticholinergics
Anticholinergic drugs[8] and deliriants:[9][10][11][12][13]
- Atropine, racemic hyoscyamine, from the deadly nightshade (Atropa belladonna)
- Hyoscyamine, the levo-isomer of atropine, from henbane (Hyoscyamus niger), mandrake (Mandragora officinarum) and the sorcerers' tree (Latua pubiflora).
- Scopolamine, from henbane and Datura species (Jimson weed)
All three acetylcholine-inhibiting chemicals can also be found in the leaves, stems, and flowers in varying, unknown amounts in Brugmansia (angel trumpets), a relative of Datura. The same is also true of many other plants belonging to subfamily Solanoideae of the Solanaceae, the alkaloids being concentrated particularly in the leaves and seeds. However, the concentration of alkaloids can vary greatly, even from leaf to leaf and seed to seed.[14][15]
Stimulants
Stimulants and cocaine-related alkaloids:[10][16]
- Cocaine, from coca plant (Erythroxylum coca)
- Ecgonine, a precursor and metabolite of cocaine
- Benzoylecgonine, a metabolite of cocaine
- Hydroxytropacocaine, from coca plant (Erythroxylum coca)
- Methylecgonine cinnamate, from coca plant (Erythroxylum coca)
Others
- Catuabines, found in catuaba, an infusion or dry extract made from Erythroxylum vaccinifolium[10][12][13]
- Scopine[10][12][13]
Synthetic analogs of tropane alkaloids also exist, such as the phenyltropanes. They are not considered to be alkaloids per definition.
Biosynthesis
The biosynthesis of the tropane alkaloids have attracted intense interest because of their high physiological activity as well as the presence of the bicyclic tropane core.[17]
Final stages in the pathway for the biosynthesis of the tropane alkaloid cocaine. Hyoscine (a.k.a: Scopolamine, INN: Hyoscine hydrobromide) is a medication used to treat motion sickness and postoperative nausea and vomiting.
Hyoscine may also refer to:
- Hyoscine butylbromide (brand name Buscopan), used for abdominal pain, renal colic, or bladder spasms
- Hyoscine methobromide Devil's Breath" is a term commonly used to describe scopolamine, a potent, naturally occurring drug derived from plants in the nightshade family, such as Brugmansia (Angel's Trumpet) or Datura stramonium (Jimsonweed).
It has gained notoriety for its reported use in criminal activities, particularly in Colombia, where it is known as burundanga, due to its ability to induce a state of extreme compliance, incapacitation, and amnesia in victims.
Key Facts About Devil's Breath (Scopolamine)
- What it is: A tropane alkaloid, specifically scopolamine or hyoscine, often found in powder form.
- Medical Use: In low, controlled doses, it is a legitimate medication listed by the World Health Organization for treating motion sickness, postoperative nausea, vomiting, and gastrointestinal spasms
Myths vs. Reality
While scopolamine is dangerous and used for crime, some aspects are sometimes sensationalized. The idea that it instantly turns people into "zombies" who can be ordered to drain their bank accounts is often exaggerated, though it does render victims highly suggestible and unable to resist. It is not easily absorbed through the skin, despite reports of it being on business cards or leaflets, and is most effective when ingested or inhaled.
Safety Advice
- Do not accept drinks, food, or cigarettes from strangers.
- Never leave your drink unattended in bars or clubs.
- Be cautious of sudden approaches by strangers offering help or trying to show you something.
If you believe you or someone else has been affected by this substance, seek immediate medical attention.
. It is commonly administered via a transdermal patch
.From Wikipedia, the free encyclopedia
For the medication used to treat abdominal pain, see Hyoscine butylbromide.
"Hyoscine" redirects here. For Other uses, see Hyoscine (disambiguation).
Not to be confused with hyoscyamine.
"Devil's Breath" redirects here. For the novel, see The Devil's Breath.
Scopolamine
Clinical data
Transderm Scop, others
Other names
Hyoscine,[1] Devil's Breath
- US DailyMed: Scopolamine
- AU: B2[2]
By mouth, transdermal, ophthalmic, subcutaneous, intravenous, sublingual, rectal, buccal, transmucosal, intramuscular
Legal status
Pharmacokinetic data
20-40%[7]
5 hours[7]
Identifiers
PubChem CID
- DL48G20X8X
- as salt: 451IFR0GXB
CompTox Dashboard (EPA)
Chemical and physical data
C17H21NO4
303.358 g·mol−1
3D model (JSmol)
Scopolamine, also known as hyoscine,[9] or Devil's Breath,[10] is a medication used to treat motion sickness[11] and postoperative nausea and vomiting.[12][1] It is also sometimes used before surgery to decrease saliva.[1] When used by injection, effects begin after about 20 minutes and last for up to 8 hours.[1] It may also be used orally and as a transdermal patch since it has been long known to have transdermal bioavailability.[1][13]
Scopolamine is in the antimuscarinic family of drugs and works by blocking some of the effects of acetylcholine within the nervous system.[1]
Scopolamine was first written about in 1881 and started to be used for anesthesia around 1900.[14][15] Scopolamine is also the main active component produced by certain plants of the nightshade family, which historically have been used as psychoactive drugs, known as deliriants, due to their antimuscarinic-induced hallucinogenic effects in higher doses.[12] In these contexts, its mind-altering effects have been utilized for recreational and occult purposes.[16][17][18]: 277–282 The name scopolamine is derived from one type of nightshade known as Scopolia, while the name "hyoscine" is derived from another type known as Hyoscyamus niger, or black henbane.[19][20] It is on the World Health Organization's List of Essential Medicines.[21]
Medical uses
Scopolamine has a number of formal uses in modern medicine where it is used in its isolated form and in low doses to treat:[22][23]
- Postoperative nausea and vomiting
- Motion sickness, including sea sickness, leading to its use by scuba divers (where it is often applied as a transdermal patch behind the ear)[24][25][26]
- Gastrointestinal spasms
- Kidney or liver spasms
- Aid in gastrointestinal radiology and endoscopy
- Irritable bowel syndrome
- Clozapine-induced drooling
- Bowel colic
- Eye inflammation[27]
It is sometimes used as a premedication, especially to reduce respiratory tract secretions in surgery, most commonly by injection.[22][23] Common side effects include sleepiness, blurred vision, dilated pupils, and dry mouth.[1] It is not recommended in people with angle-closure glaucoma or bowel obstruction.[1] Whether its use during pregnancy is safe remains unclear, and use during breastfeeding is still cautioned by health professionals and manufacturers of the drug.[28]
Scopolamine can be taken by mouth, subcutaneously, in the eye, and intravenously, as well as via a transdermal patch.[29]
Breastfeeding
Scopolamine enters breast milk by secretion. Although no human studies exist to document the safety of scopolamine while nursing, the manufacturer recommends that caution be taken if scopolamine is administered to a breastfeeding woman.[28]
Adverse effects
Adverse effect incidence:[6][30][31][32]
Uncommon (0.1–1% incidence) adverse effects include:
- Dry mouth
- Anhidrosis (reduced ability to sweat to cool off)
- Tachycardia (usually occurs at higher doses and is succeeded by bradycardia)
- Bradycardia
- Urticaria (hives)
- Pruritus (itching)
Rare (<0.1% incidence) adverse effects include:
- Constipation
- Urinary retention
- Hallucinations
- Agitation
- Delirium
- Restlessness
- Seizures
Unknown frequency adverse effects include:
- Anaphylactic shock or reactions
- Dyspnea (shortness of breath)
- Rash
- Erythema
- Other hypersensitivity reactions
- Blurred vision
- Mydriasis (dilated pupils)
- Drowsiness
- Dizziness
- Somnolence
Overdose
Physostigmine, a cholinergic drug that readily crosses the blood–brain barrier, has been used as an antidote to treat the central nervous system depression symptoms of a scopolamine overdose.[33] Other than this supportive treatment, gastric lavage and induced emesis (vomiting) are usually recommended as treatments for oral overdoses.[32] The symptoms of overdose include:[31][32]
- Tachycardia
- Arrhythmia
- Blurred vision
- Photophobia
- Urinary retention
- Drowsiness or paradoxical reaction, which can present with hallucinations
- Cheyne–Stokes respiration
- Dry mouth
- Skin reddening
- Inhibition of gastrointestinal motility
Pharmacology
Pharmacodynamics
The pharmacological effects of scopolamine are mediated through the drug's competitive antagonism of the peripheral and central muscarinic acetylcholine receptors. Scopolamine acts as a nonspecific muscarinic antagonist at all four (M1, M2, M3, and M4) receptor sites.[34][35]
In doses higher than intended for medicinal use, the hallucinogenic alteration of consciousness, as well as the delirium in particular, are tied to the compound's activity at the M1 muscarinic receptor. M1 receptors are located primarily in the central nervous system and are involved in perception, attention, and cognitive functioning. Delirium is associated solely with the antagonism of postsynaptic M1 receptors; currently no other receptor subtypes have been implicated.[36]
Peripheral muscarinic receptors are part of the autonomic nervous system. M2 receptors are located in the brain and heart, M3 receptors are in salivary glands and M4 receptors are in the brain and lungs.[36] Due to the drug's inhibition of various signal transduction pathways, the decrease in acetylcholine signaling is what leads to many of the cognitive deficits, mental impairments, and delirium associated with psychoactive doses. Medicinal effects appear to mostly be tied to activation of the peripheral receptors and only from marginal decreases in acetylcholine signaling.[37]
Although often broadly referred to as simply being 'anticholinergic', antimuscarinic would be more specific and accurate terminology to use for scopolamine, as, for example, it is not known to block nicotinic receptors.[36]
Pharmacokinetics
Scopolamine undergoes first-pass metabolism and about 2.6% is excreted unchanged in urine. It has a bioavailability of 20–40%, reaches peak plasma concentration in about 45 minutes, and in healthy subjects has an average half-life of 5 hours (observed range 2–10 hours).[7] Scopolamine is primarily metabolized by the CYP3A4 enzyme, and grapefruit juice decreases metabolism of scopolamine, consequently increasing plasma concentration.[8]
Chemistry
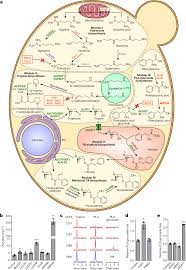
Biosynthesis in plants
Scopolamine is among the secondary metabolites of plants from the Solanaceae (nightshade) family of plants, including henbane (Hyoscyamus niger), jimson weed (Datura), angel's trumpets (Brugmansia), deadly nightshade (Belladonna), mandrake (Mandragora officinarum), and corkwood (Duboisia).[38][19]
The biochemistry of tropane class compounds
The biosynthesis of scopolamine begins with the decarboxylation of L-ornithine to putrescine by ornithine decarboxylase. Putrescine is methylated to N-methylputrescine by putrescine N-methyltransferase.[39]
A putrescine oxidase that specifically recognizes methylated putrescine catalyzes the deamination of this compound to 4-methylaminobutanal, which then undergoes a spontaneous ring formation to N-methyl-pyrrolium cation. In the next step, the pyrrolium cation condenses with acetoacetic acid yielding hygrine. No enzymatic activity could be demonstrated to catalyze this reaction. Hygrine further rearranges to tropinone.[39]
Subsequently, tropinone reductase I converts tropinone to tropine, which condenses with phenylalanine-derived phenyllactate to littorine. A cytochrome P450 classified as Cyp80F1[40] oxidizes and rearranges littorine to hyoscyamine aldehyde. In the final step, hyoscyamine undergoes epoxidation catalyzed by 6β-hydroxyhyoscyamine epoxidase yielding scopolamine.[39]